
The use of photo-ionisation detector (PID) in recent years has become increasingly common. For the detection of volatile organic compounds such as benzene or toluene no other common sensor type comes close to detecting the gas at the part per million (or even part per billion) level required, and the broad range of organic compounds it detects in a compact and reliable format, make it ideal for the application. The PID sensor only works however because of a strange property of light (in this case ultra-violet (UV) light). The property is known in quantum mechanics as the “wave-particle duality”.
The “wave-particle duality” means that light behaves in two different ways at the same time. It moves through space (at the speed of…er…light!) as a wave, just like a wave on the ocean moving relentlessly to shore, but the energy that is carried by the wave is delivered like a particle, or packet of energy – as if it were a box delivered by Amazon!
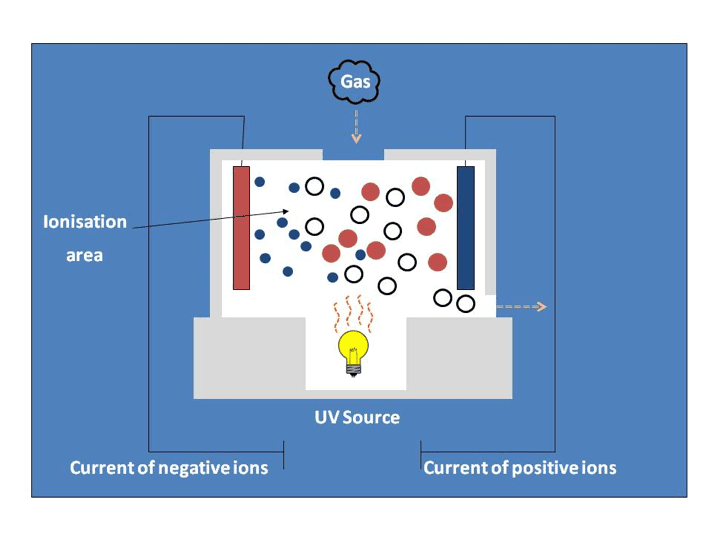
In a PID sensor the UV light travels quickly towards the target gas, and when it gets there it delivers a whack of energy, just like a boxer gliding across the ring and whomping their opponent. The whack of energy is enough to break the gas (the white ball in the diagram) into two pieces, a bit like knocking out the opponents tooth. The smaller (tooth) piece is a negatively charged electron (blue in the diagram), and the larger remaining piece is a positively charged ion (red in the diagram) and once separated and charged they can create a current by moving to plates of different polarities inside the sensor. This current is used to make the measurement of the gas concentration, literally, the more gas, the more ions, the more current. Simple.
Also as with boxing there are different categories of PID based on the energy of the lamp, 9.8eV (electron volts) might be a featherweight, 10.6eV the middle weight, and 11.7eV the heavy weight, and each has more suitable targets. Materials are detected according to their ionisation potential (IP), this is a measure of the amount of energy required to ionise the material, or put another way, how hard you have to hit the boxer to knock his tooth out! For a material like isobutylene (the most comment calibration gas used for PIDs) the ionisation potential is 9.24eV meaning it is easily “beaten up” at any level, and therefore an easy and reliable gas to use as a standard. A great many VOC’s have an IP of greater than 9.8eV, but less than 10.6eV making the 10.6eV lamp the most commonly used in instrumentation (as it delivers a broad measurement with a relative robustness which the 11.7eV lamp does not have).
Benzene is a significant inherent component of crude oil and thus oil products and can comprise up to 15% of some crudes. It is a colourless liquid with a sweet smell, and is the simplest of the ‘aromatic’ compounds. Long-term exposure to benzene can lead to
- Acute Myeloid Leukaemia, (a type of cancer that affects the blood)
- Secondary Aplastic Anaemia
- Damage to the reproductive system
- Long-term exposure to benzene can also cause other types of leukaemia and severe anaemia.
- 1ppm exposure over 40 years can lead to 1.4 -14 excess deaths per 1000
- 10ppm exposure over 40 years can lead to 44-152 excess deaths per 1000
Pretty nasty stuff!
For a material like benzene with an IP of 9.24eV using the 10.6 eV lamp it will give a strong response, but by using a lamp with a lower output such as 9.8eV it is possible to drastically reduce the number of other gases which will respond such that benzene in many environments can be determined selectively. It is also possible to add further screening tubes to single benzene out completely, this reduces the measurement to a “single shot” device, but it is a very effective way to detect the gas.

Mohammed Ali famously stated the he floats like a butterfly, stings like a bee, and the floaty wave sucker punch that the PID delivers shows why it is the Mohammed Ali of the gas detector world. As with boxing, it is really a matter of opinion whether that makes it “the Greatest”, but it is certainly more than a match for benzene!
